Exploring the Maxwell-Boltzmann Distribution with AIMS
The Maxwell-Boltzmann distribution describes how atomic and molecular speeds are distributed in a gas. AIMS lets you see it, vary it with temperature, and compare it across molecules.
The distribution of speeds of atoms and molecules obeys the Maxwell-Boltzmann distribution:
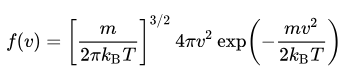
where v is the speed, f(v) is the probability density function, kB is the Boltzmann constant, T is the temperature, and m is the mass of the particle (atom or molecule). If statistical mechanics is not in your wheelhouse, you can play with an interactive molecular dynamics simulation of some argon atoms below to visualize this distribution.
Now, AIMS is not just a visualization tool. It is a complete molecular design, simulation, and analysis environment, which allows you to make deeper scientific inquiry. The following provides a list of things that you can do with AIMS to explore the Maxwell-Boltzmann speed distribution further.
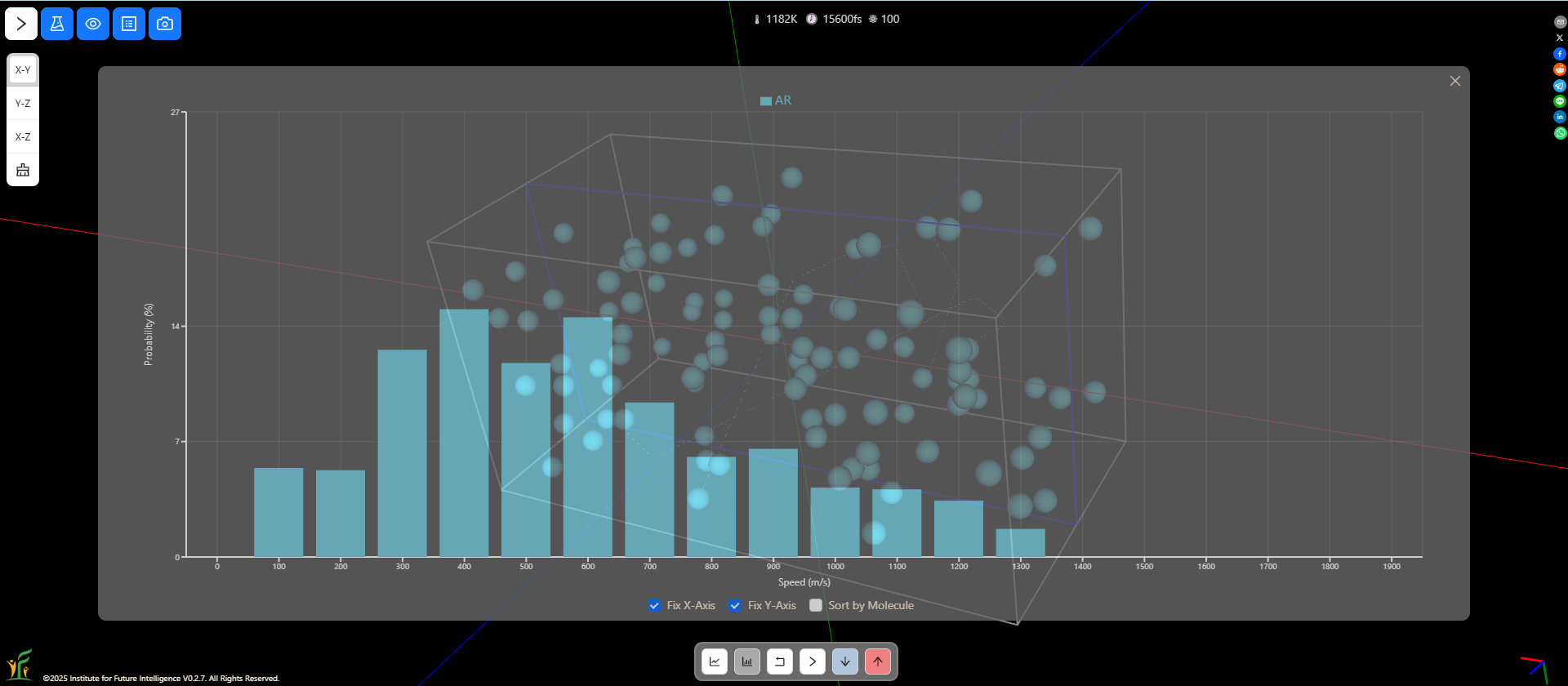
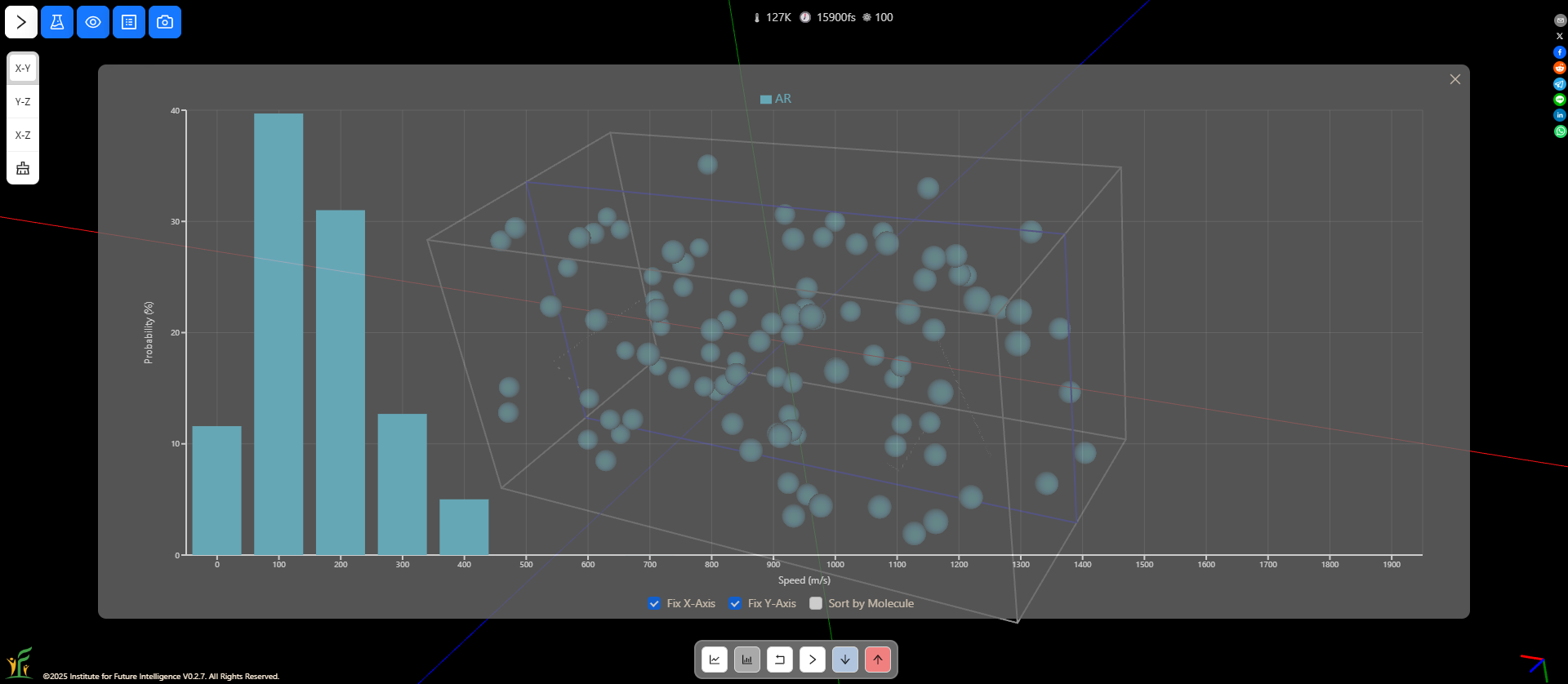
Speed distributions at different temperatures
You can change the temperature in a molecular dynamics simulation and observe how the speed distribution varies as a result. At a higher temperature, there are more molecules that move at higher speeds.
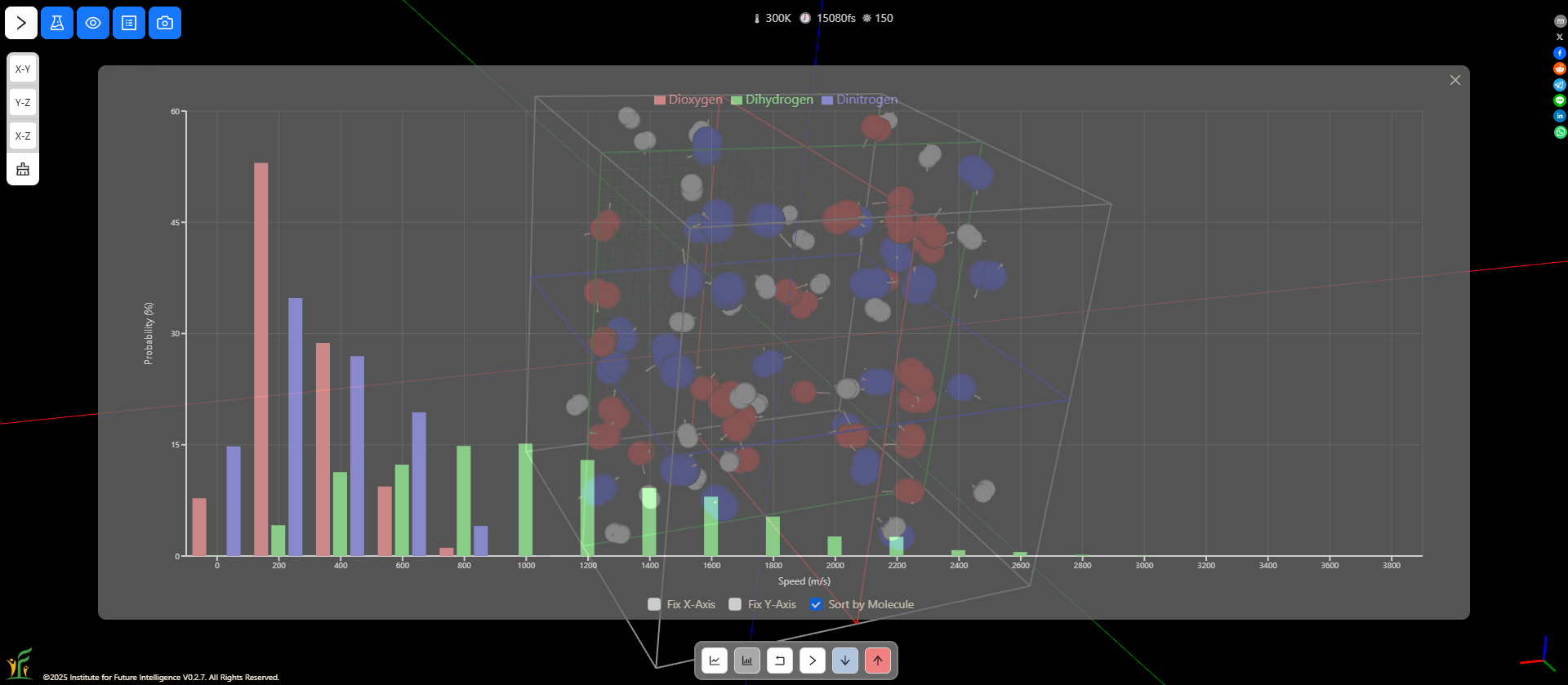
Speed distributions of different types of molecules
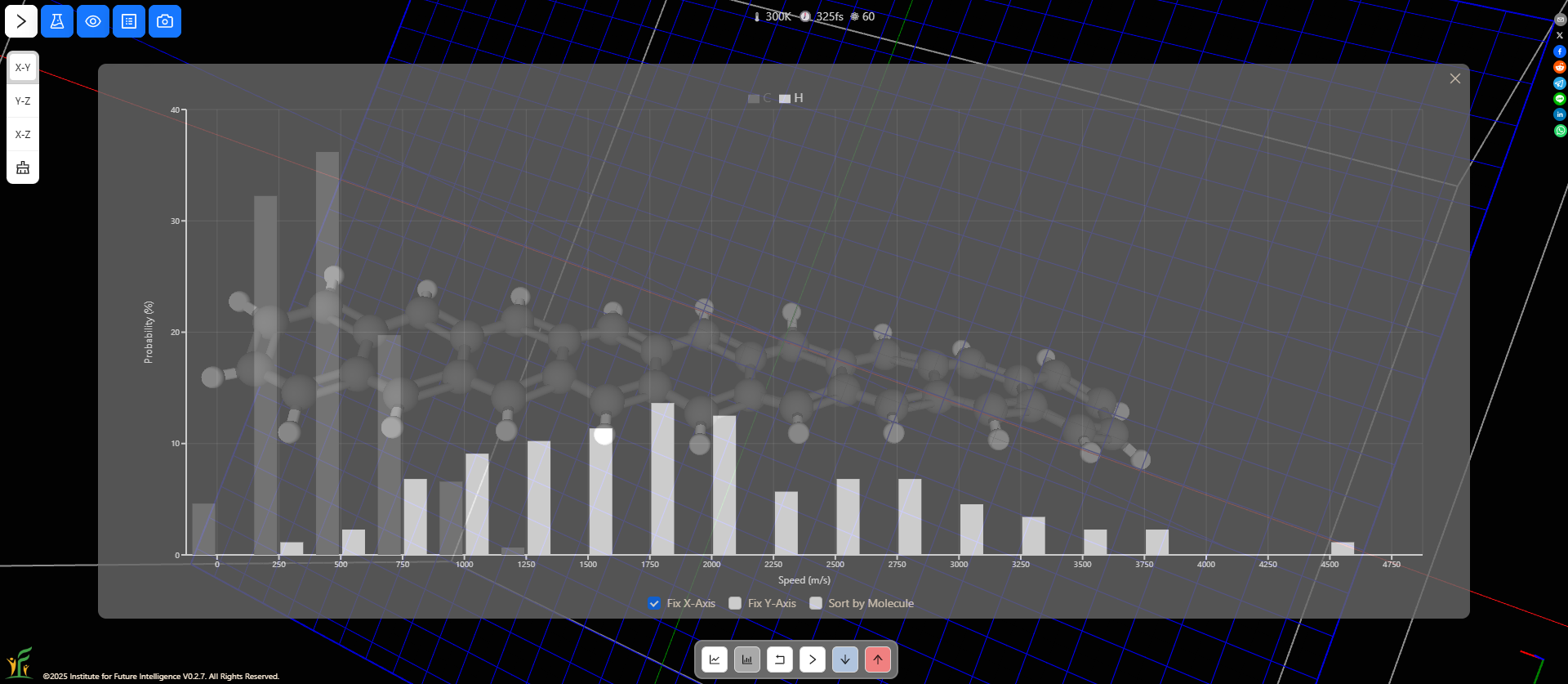
AIMS allows you to sort the speeds by atoms or molecules. You can compare the distributions of different types of molecules as shown in the following screenshot. Molecules with less mass on average move faster than those with more mass.
Speed distributions of different atoms of molecules
What about different types of atoms that are part of a molecule? Do they also obey the Maxwell-Boltzmann distribution? You can easily check this out with AIMS. The following screenshot shows that the hydrogen atoms of a long hydrocarbon molecule, on average, move at a higher speed than the carbon atoms, even though the two types are bonded.